Get Free Delivery With No Minimum Order
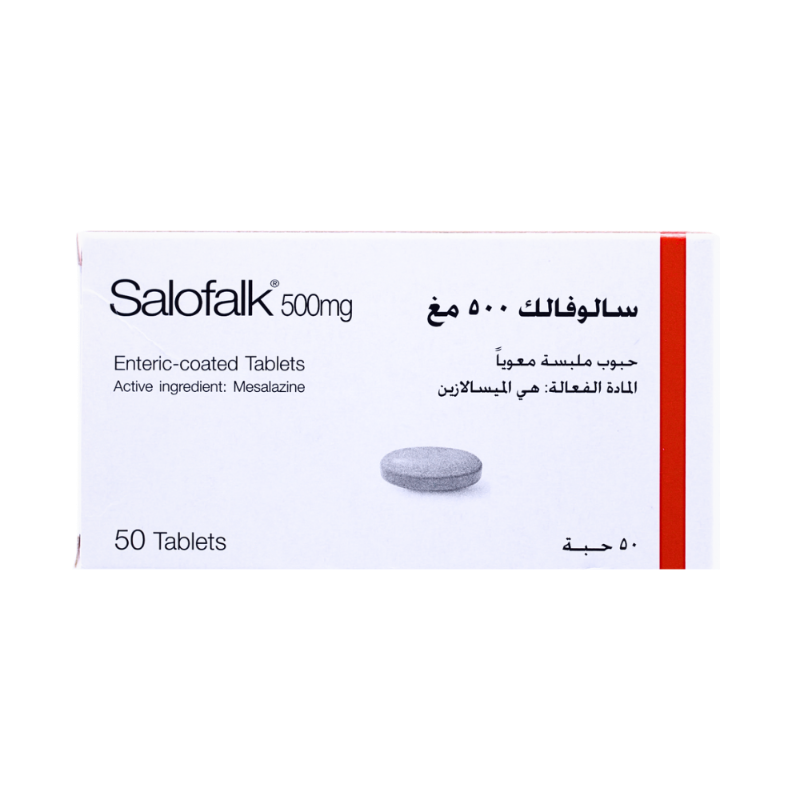
SALOFALK 500MG TAB
- Sku : I-005728
Key features
SALOFALK 500MG TAB is a gastro-resistant tablet formulation containing mesalazine 500 mg. Mesalazine acts topically on the intestinal mucosa to reduce inflammation, in part by inhibiting cyclooxygenase and lipoxygenase pathways and decreasing prostaglandin and leukotriene production. It is indicated for treatment of acute episodes of mild to moderate ulcerative colitis, maintenance of remission in ulcerative colitis, and treatment of acute attacks of Crohn's disease. Available on prescription in packs of 50 gastro-resistant tablets.- Brand: SALOFALK
- Active Ingredient: MESALAZINE 500mg
- Strength: 500mg
- Dosage Form: Gastro-resistant tablet
- Pack Size: 50 Tablets
- Route: Oral use
- Prescription Status: Prescription
- Therapeutic Class: Gastrointestinal
- Pharmacological Group: Antidiarrheals & Anti-inflammatory
- Drug Class: Aminosalicylate Anti-inflammatory Agent (5-ASA)
- Manufacturer: LOSAN PHARMA GMBH
- Country of Origin: Germany
- SFDA Registration No.: 1705222031
- Shelf Life: 36 months
- Storage: do not store above 30°c
- Gi Condition: Inflammatory Bowel Disease
Indications
Approved Uses
Treatment of acute episodes of mild to moderate ulcerative colitis, Maintenance of remission of ulcerative colitis, Treatment of acute attacks of Crohn's disease.
Dosage & Administration
Dosing by Condition
Ulcerative colitis (active): 1.5-3 g/day in divided doses (some guidelines allow up to 4-4.8 g/day depending on formulation/severity). Ulcerative colitis (maintenance): typically 1.5 g/day in divided doses (range commonly 1-2 g/day; some patients may require up to 3 g/day). Crohn’s disease: mesalazine has limited benefit; if used, doses around 2-4 g/day have been studied, but it is not routinely recommended for induction/maintenance in most guidelines.
Initial Dose
For acute ulcerative colitis: 1.5g to 3g daily, for example one 500mg tablet three times daily, which can be increased.
Maintenance Dose
500mg two to three times daily (1-1.5g/day)
Maximum Dose
3g/day for active disease
Children's Dosage
Children 5 years and older: weight-based dosing
Dose Adjustment Notes
No routine dose adjustment is recommended, but use with caution and monitor closely in renal impairment; contraindicated/avoid in severe renal impairment. Use with caution in hepatic impairment (avoid in severe hepatic impairment).
How to Take
Swallow the gastro-resistant tablets whole with plenty of water; do not crush, chew, or break. Take in divided doses as prescribed; may be taken with or without food (follow product-specific directions if provided).
Side Effects
Common Side Effects
Headache, nausea, abdominal pain, diarrhea, flatulence, vomiting, dyspepsia, rash
Safety & Warnings
Contraindications
Hypersensitivity to mesalazine/salicylates or excipients; severe renal impairment; severe hepatic impairment.
Warnings & Precautions
Baseline and periodic renal function monitoring is essential; monitor liver function and consider CBC (especially if on thiopurines or if symptoms suggest blood dyscrasia); stop and assess if acute intolerance syndrome occurs (cramps, acute abdominal pain, bloody diarrhea, fever); use caution in asthma/pulmonary disease and in patients with prior peptic ulcer disease; counsel to report rash, chest pain, dyspnea, or reduced urine output promptly.
Age Restriction
Not recommended in children <6 years (limited experience); use in pediatrics only under specialist supervision with weight-based dosing.
Drug Interactions
Interaction Severity
MAJOR: Azathioprine/6-mercaptopurine/thioguanine (additive myelosuppression-monitor CBC closely). MODERATE: NSAIDs (increased risk of renal adverse effects), methotrexate (possible increased toxicity-monitor), warfarin (variable effect on anticoagulation-monitor INR). MINOR/FORMULATION-RELATED: agents that markedly lower intestinal pH (may affect pH-dependent release for some products).
Special Populations
Pregnancy
Caution
Children
Children 5 years and older: weight-based dosing
Storage & Patient Advice
Stopping the Medicine
Do not stop without prescriber advice; stopping can precipitate relapse-maintenance therapy is often long-term, and any change should be clinician-directed (no routine taper required).
Patient Counseling
Swallow tablets whole with water; do not crush/chew (gastro-resistant). Take regularly as prescribed and continue even when symptoms improve. Attend scheduled renal (and other) blood/urine monitoring. Seek care urgently for signs of hypersensitivity/intolerance (worsening bloody diarrhea/cramps, fever), unexplained bruising/bleeding or sore throat (possible blood dyscrasia), or reduced urine output/flank pain (kidney injury).
Monitoring Requirements
Baseline and periodic renal function (serum creatinine/eGFR and urinalysis); consider baseline and periodic CBC and liver enzymes, especially early in therapy and in at-risk patients; increase monitoring frequency if renal/hepatic impairment or concomitant nephrotoxic/myelosuppressive drugs.
Pharmacology
Mechanism of Action
Mechanism is not fully defined; mesalazine acts topically on intestinal mucosa to reduce inflammation, in part via inhibition of cyclooxygenase and lipoxygenase pathways and downstream prostaglandin/leukotriene production (and other local anti-inflammatory effects).
Onset of Action
Symptom improvement is often seen within 2-4 weeks; full benefit for induction may take up to 6-8 weeks.
Duration of Effect
Effect is maintained only with continued therapy; remission maintenance requires ongoing dosing, and relapse risk increases after discontinuation.
Half-Life
Mesalazine: ~0.5-2 hours; N-acetyl-mesalazine: ~5-10 hours (values vary by formulation and sampling).
Bioavailability
Approximately 20-30% systemic absorption (varies by formulation), with the remainder acting locally in the gut.
Metabolism
Primarily acetylated in the intestinal mucosa and liver to N-acetyl-5-aminosalicylic acid; not meaningfully CYP450-mediated.
Excretion
Unabsorbed drug is excreted in feces; absorbed mesalazine is largely converted to N-acetyl-mesalazine and eliminated mainly via the kidneys (urine).
Product Information
Available Dosage Forms
Gastro-resistant tablets, prolonged-release tablets, granules, rectal suspension (enema), suppositories, rectal foam
Composition per Dose
Each gastro-resistant tablet: 500mg mesalazine (5-aminosalicylic acid).
Generic Availability
Yes
OTC Alternatives
No OTC alternative
Gi Condition
Inflammatory Bowel Disease
Legal Disclaimer - Al Mujtama Pharmacy
The product information provided is derived from verified pharmaceutical references and is intended for general health education only. It is not a substitute for professional medical advice, diagnosis, or treatment.
Al Mujtama Pharmacy assumes no legal or medical liability for:
- Any therapeutic decision made based on the information displayed without consulting a licensed physician or pharmacist
- Any discrepancy between the information provided and the product's package insert or SFDA guidelines
- Any misuse of medication resulting from personal interpretation of the content displayed
Important notice: Drug formulations and instructions may vary between production batches. Always rely on the leaflet included inside the product packaging you have, and consult your pharmacist or physician before starting, adjusting, or discontinuing any medication.
By using this content, you acknowledge that you have read this disclaimer and agree that Al Mujtama Pharmacy bears no liability arising from reliance on this information as a substitute for direct medical consultation.
Your health is a trust - always consult your doctor first.
-1744229570.gif)