Get Free Delivery With No Minimum Order





CLARISPRAY NASAL SPRAY DECONGESTANT ORIGINAL 15ML
- Sku : I-033204
Key features
CLARISPRAY Nasal Spray Decongestant Original 15 ml from CLARICARE contains oxymetazoline 0.5 mg/ml as the active ingredient. As an imidazoline-derived alpha-adrenergic agonist, it produces local vasoconstriction of the nasal mucosa to reduce swelling and open nasal passages. It provides temporary relief of nasal congestion due to the common cold, hay fever or other upper respiratory allergies, and relieves sinus congestion and pressure. Available over the counter as a 15 ml nasal spray.- Brand: CLARICARE
- Active Ingredient: OXYMETAZOLINE 0.5mg/ml
- Strength: 0.5mg/ml
- Dosage Form: Nasal spray
- Pack Size: 15 ml
- Route: Nasal use
- Prescription Status: OTC
- Drug Class: Topical nasal decongestant; imidazoline-derived sympathomimetic alpha-adrenergic agonist.
- Manufacturer: Delpharm Montréal Inc
- Country of Origin: Canada
- SFDA Registration No.: 1307233870
- Shelf Life: 30 months
- Storage: do not store above 30°c
- Symptom Target: Congestion
- Sedating: No
Indications
Approved Uses
Temporary relief of nasal congestion due to the common cold, hay fever, or other upper respiratory allergies (allergic rhinitis), and sinus congestion and pressure.
Off-Label Uses
Adjunct for control of anterior epistaxis and to reduce nasal mucosal bleeding during nasal procedures (topical vasoconstrictor use).
Dosage & Administration
Dosing by Condition
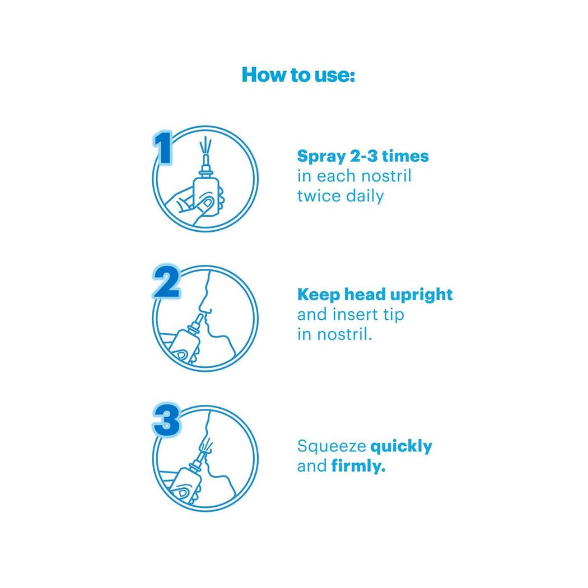
Nasal congestion (adults and children ≥6 years): 2-3 sprays in each nostril every 10-12 hours as needed; do not exceed 2 doses in 24 hours; do not use longer than 3 consecutive days.
Dose Adjustment Notes
No routine renal/hepatic dose adjustment is recommended for typical intranasal use; limit use to a maximum of 3 consecutive days to avoid rebound congestion.
How to Take
Blow nose gently before use; keep head upright or slightly forward (do not tilt back); insert nozzle into one nostril while occluding the other; spray while breathing in gently (do not inhale deeply); repeat for other nostril; wipe nozzle and replace cap after use; do not share.
Side Effects
Common Side Effects
Transient nasal burning/stinging, dryness/irritation, sneezing; headache may occur; rebound congestion (rhinitis medicamentosa) with use beyond 3 days.
Safety & Warnings
Contraindications
Contraindications: hypersensitivity to oxymetazoline/excipients; concomitant use of MAO inhibitors or within 14 days of stopping; (often also listed) atrophic/dry rhinitis-while angle-closure glaucoma and hyperthyroidism are typically precautions rather than absolute contraindications.
Warnings & Precautions
Do not use longer than 3 consecutive days; use with caution/seek medical advice in hypertension/heart disease, hyperthyroidism, diabetes, prostatic hypertrophy/urinary retention, and narrow-angle glaucoma; avoid eyes and do not share the spray.
Age Restriction
Not recommended for children under 6 years unless directed by a doctor; use age-appropriate pediatric strength/products for younger children.
Drug Interactions
Drug Interactions
Clinically significant interactions: MAO inhibitors (and within 14 days) and tricyclic antidepressants (increased pressor/sympathomimetic effects); additive effects with other sympathomimetics; caution with antihypertensives/beta-blockers due to potential BP/HR effects.
Special Populations
Pregnancy
Consult Doctor
Breastfeeding
Consult Doctor
Elderly
Standard adult dosing; use with caution due to increased risk of cardiovascular effects and prostatic hypertrophy
Kidney Impairment
No dose adjustment recommended/required for renal impairment for intranasal oxymetazoline when used as directed.
Liver Impairment
No dose adjustment recommended/required for hepatic impairment for intranasal oxymetazoline when used as directed.
Storage & Patient Advice
Missed Dose
Usually used as needed; if using on a schedule and a dose is missed, use when remembered unless it is close to the next dose-then skip; do not double doses and do not exceed 2 doses in 24 hours.
Stopping the Medicine
Safe to stop at any time; if used longer than 3 days, rebound congestion (rhinitis medicamentosa) may occur and stopping plus saline/supportive care is advised.
Overdose
Overdose/excess use or ingestion (especially in children) can cause serious CNS and cardiovascular effects (e.g., marked drowsiness/CNS depression, bradycardia, hypotension or hypertension, hypothermia, respiratory depression/coma, or agitation/tachycardia); seek urgent medical care/poison center and treat supportively.
Patient Counseling
Use only in the nose and follow labeled dose (2-3 sprays/nostril every 10-12 hours; max 2 doses/day); do not use longer than 3 consecutive days (rebound congestion risk); avoid sharing the spray; keep out of eyes; seek advice before use if you have hypertension/heart disease, hyperthyroidism, diabetes, or glaucoma, or if taking MAOIs; consult a clinician if symptoms persist/worsen or if significant palpitations, headache, or BP elevation occurs.
Monitoring Requirements
No routine monitoring for most patients; consider monitoring blood pressure/heart rate in patients with cardiovascular disease, hypertension, hyperthyroidism, diabetes, or when overuse is suspected.
Pharmacology
Mechanism of Action
Alpha-adrenergic agonist (primarily alpha-1 with some alpha-2 activity) causing vasoconstriction of nasal mucosal vessels, reducing edema and nasal congestion.
Onset of Action
Within 5-10 minutes.
Duration of Effect
Up to 10-12 hours
Bioavailability
Minimal systemic absorption with recommended intranasal use; systemic exposure increases with excessive dosing, prolonged use, or mucosal damage.
Excretion
Excreted mainly in urine with a smaller portion in feces (systemic absorption is low after intranasal use).
Protein Binding
Not well established/variable in standard clinical references (treat as unknown for practical labeling-level summaries).
Product Information
Available Dosage Forms
Nasal spray, nasal drops, eye drops, topical cream.
Composition per Dose
Each spray (approximately 0.1 mL): 0.05 mg oxymetazoline hydrochloride (equivalent to 0.5 mg/mL concentration)
Generic Availability
Yes
OTC Alternatives
Xylometazoline nasal spray, saline nasal spray, pseudoephedrine oral decongestant
Symptom Target
Congestion
Sedating
No
Legal Disclaimer - Al Mujtama Pharmacy The product information provided is derived from verified pharmaceutical references and is intended for general health education only. It is not a substitute for professional medical advice, diagnosis, or treatment. Al Mujtama Pharmacy assumes no legal or medical liability for: Any therapeutic decision made based on the information displayed without consulting a licensed physician or pharmacist Any discrepancy between the information provided and the product's package insert or SFDA guidelines Any misuse of medication resulting from personal interpretation of the content displayed Important notice: Drug formulations and instructions may vary between production batches. Always rely on the leaflet included inside the product packaging you have, and consult your pharmacist or physician before starting, adjusting, or discontinuing any medication. By using this content, you acknowledge that you have read this disclaimer and agree that Al Mujtama Pharmacy bears no liability arising from reliance on this information as a substitute for direct medical consultation. Your health is a trust - always consult your doctor first.-1744229570.gif)