Get Free Delivery With No Minimum Order
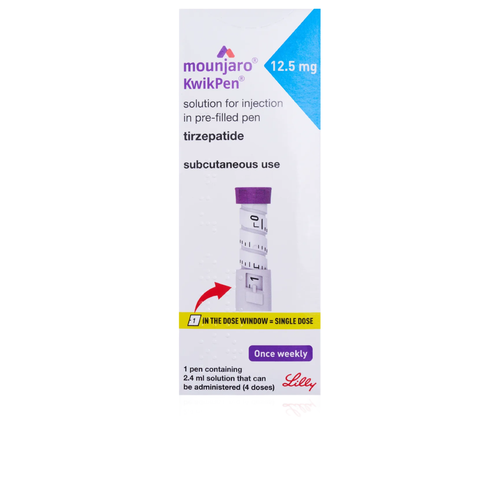
MOUNJARO KWIKPEN 12.5MG/0.6ML 3ML X1
- Sku : I-032844
Key features
MOUNJARO KwikPen 12.5 mg is a pre-filled solution for injection containing the active ingredient tirzepatide 12.5 mg. It acts as a dual GIP/GLP-1 receptor agonist, enhancing glucose-dependent insulin secretion, suppressing glucagon release and slowing gastric emptying to lower blood glucose and reduce appetite. It is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus. Supplied as a single 3 mL pre-filled KwikPen.- Brand: MOUNJARO
- Active Ingredient: TIRZEPATIDE 12.5mg
- Strength: 12.5mg
- Dosage Form: Solution for injection in pre-filled pen
- Pack Size: 3 ml
- Route: Subcutaneous use
- Prescription Status: Prescription
- Therapeutic Class: Glucose-dependent Insulinotropic Polypeptide Receptor Agonist [EPC], GLP-1 Receptor Agonist [EPC]
- Pharmacological Group: G-Protein-linked Receptor Interactions [MoA], Glucagon-like Peptide-1 (GLP-1) Agonists [MoA]
- Drug Class: Dual GIP/GLP-1 Receptor Agonist (Twincretin)
- Manufacturer: Eli Lilly Italia SpA
- Country of Origin: Italy
- SFDA Registration No.: 1208245749
- Shelf Life: 24 months
- Storage: store in a refrigerator (2°c - 8°c)
- Diabetes Type: Type 2
Indications
Approved Uses
As an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Off-Label Uses
Off-label: weight management/obesity (where not specifically approved under the Mounjaro indication), and investigational/off-label metabolic conditions such as NAFLD/NASH or PCOS-related metabolic dysfunction.
Dosage & Administration
Dosing by Condition
Type 2 diabetes mellitus (adults): 2.5 mg SC once weekly for 4 weeks, then 5 mg once weekly; if additional control needed, increase by 2.5 mg every ≥4 weeks to maintenance 5-15 mg once weekly (max 15 mg weekly).
Initial Dose
2.5 mg subcutaneously once weekly for 4 weeks (this dose is for treatment initiation and not for glycemic control).
Maintenance Dose
5 mg to 15 mg subcutaneously once weekly
Maximum Dose
15 mg subcutaneously once weekly (maximum maintenance dose).
Children's Dosage
Approved for pediatric patients 10 years of age and older with type 2 diabetes mellitus.
Dose Adjustment Notes
Increase dose in 2.5 mg increments after at least 4 weeks on the current dose based on tolerability/need; if not tolerated, consider delaying escalation. The weekly dosing day can be changed if ≥72 hours between doses. No routine dose adjustment is required for renal or hepatic impairment, but monitor closely if severe GI reactions occur (risk of dehydration/AKI).
How to Take
For subcutaneous injection only: Inject tirzepatide once weekly in the abdomen, thigh, or upper arm; rotate injection sites. Administer at any time of day, with or without meals. Do not shake. The day of weekly administration may be changed if the time between doses is at least 72 hours.
How to Prepare
Ready-to-use pre-filled pen (no reconstitution); visually inspect the solution before injection-use only if clear to slightly opalescent and colorless to slightly yellow, and do not use if discolored or contains particles.
Side Effects
Common Side Effects
Nausea, diarrhea, decreased appetite, vomiting, constipation, dyspepsia/indigestion, and abdominal pain; injection-site reactions may occur. Hypoglycemia risk increases when used with insulin or a sulfonylurea.
Side Effect Frequency
Very common (≥10%): nausea, diarrhea. Common (1%-<10%): decreased appetite, vomiting, constipation, abdominal pain, injection-site reactions, fatigue, eructation, flatulence, gastroesophageal reflux; hypoglycemia when used with insulin or sulfonylurea.
Safety & Warnings
Contraindications
Personal or family history of medullary thyroid carcinoma (MTC), Multiple Endocrine Neoplasia syndrome type 2 (MEN 2), known serious hypersensitivity to tirzepatide or its excipients.
Warnings & Precautions
Boxed warning/contraindication for MTC/MEN2; pancreatitis risk (discontinue if suspected); hypoglycemia risk with insulin/secretagogues (consider dose reduction); acute kidney injury risk from dehydration due to GI adverse effects; severe GI disease/gastroparesis-use not recommended; diabetic retinopathy complications may worsen with rapid glycemic improvement; acute gallbladder disease; serious hypersensitivity reactions.
Age Restriction
Approved for use in patients 10 years of age and older with type 2 diabetes.
Drug Interactions
Drug Interactions
Insulin/insulin secretagogues: increased hypoglycemia risk (consider dose reduction). Delayed gastric emptying may affect absorption of oral drugs; oral contraceptives may have reduced exposure-use non-oral or add barrier contraception for 4 weeks after initiation and 4 weeks after each dose escalation.
Interaction Severity
MAJOR: Concomitant insulin or sulfonylureas (increased hypoglycemia risk-consider dose reduction and monitor). MODERATE: Oral hormonal contraceptives (delayed gastric emptying may reduce exposure-use non-oral or add barrier contraception for 4 weeks after initiation and after each dose increase).
Food Interaction
No food restrictions; administer with or without meals.
Special Populations
Pregnancy
Consult Doctor
Breastfeeding
Consult Doctor
Children
Approved for pediatric patients 10 years of age and older with type 2 diabetes mellitus.
Elderly
Standard adult dosing; no dose adjustment required based on age alone. Use with caution in elderly patients prone to dehydration from GI side effects; monitor renal function
Kidney Impairment
No dose adjustment required for renal impairment, including end-stage renal disease; monitor renal function if severe GI reactions occur (risk of dehydration/AKI).
Liver Impairment
No dose adjustment required in hepatic impairment.
Storage & Patient Advice
Storage Conditions
store in a refrigerator (2°c - 8°c)
Preparation Instructions
Ready-to-use pre-filled pen (no reconstitution); visually inspect the solution before injection-use only if clear to slightly opalescent and colorless to slightly yellow, and do not use if discolored or contains particles.
Missed Dose
If a dose is missed, administer as soon as possible within 4 days (96 hours). If more than 4 days have passed, skip the missed dose and take the next dose on the regularly scheduled day. Do not take two doses within 3 days (72 hours).
Stopping the Medicine
Can be stopped without tapering, but glycemic control and weight benefits may diminish; consult prescriber before stopping.
Overdose
Likely symptoms: severe nausea/vomiting and hypoglycemia (especially with insulin/secretagogues). Management: supportive care, treat hypoglycemia with glucose, maintain hydration/monitor renal function; no specific antidote; seek urgent medical care.
Patient Counseling
Inject once weekly on the same day each week SC in abdomen/thigh/upper arm and rotate sites; may take with or without meals; do not shake. If changing the dosing day, ensure ≥72 hours between doses. Missed dose: take within 96 hours or skip. Common GI effects (nausea/diarrhea/vomiting/constipation) are expected early-eat smaller meals and stop when full. If using insulin or a sulfonylurea, monitor for hypoglycemia. Seek urgent care for severe/persistent abdominal pain (pancreatitis) or symptoms of gallbladder disease. Counsel on boxed warning risk of thyroid C-cell tumors and to report neck mass/hoarseness/dysphagia; avoid in patients with personal/family history of MTC or MEN2. If using oral contraceptives, use non-oral or add barrier contraception for 4 weeks after starting and after each dose increase. Store refrigerated 2-8°C; do not freeze.
Monitoring Requirements
Monitor glycemic control (SMBG as appropriate and HbA1c periodically), weight, and tolerability; monitor for pancreatitis symptoms and gallbladder disease; monitor renal function in patients with severe GI adverse reactions (dehydration risk); if pre-existing diabetic retinopathy, monitor for progression; monitor for hypoglycemia when used with insulin/sulfonylurea.
Pharmacology
Mechanism of Action
A dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. It increases insulin secretion, reduces glucagon secretion, and slows gastric emptying to lower blood glucose levels and reduce appetite.
Duration of Effect
Approximately 1 week (supports once-weekly dosing).
Half-Life
Approximately 5 days (120 hours)
Metabolism
Proteolytic cleavage of the peptide backbone, beta-oxidation of the C18 fatty diacid moiety, and amide hydrolysis; not metabolized via CYP450 enzymes
Excretion
Elimination occurs via proteolytic degradation to amino acids/peptide fragments; intact tirzepatide is not expected to be excreted unchanged, and specific renal vs fecal excretion percentages are not characterized in labeling.
Product Information
Available Dosage Forms
Solution for injection for subcutaneous use in a pre-filled pen (KwikPen).
Composition per Dose
Each 0.5 mL dose: 12.5 mg tirzepatide (as free peptide) in solution for injection
Generic Availability
No
OTC Alternatives
No OTC alternative
Diabetes Type
Type 2
Legal Disclaimer - Al Mujtama Pharmacy
The product information provided is derived from verified pharmaceutical references and is intended for general health education only. It is not a substitute for professional medical advice, diagnosis, or treatment.
Al Mujtama Pharmacy assumes no legal or medical liability for:
- Any therapeutic decision made based on the information displayed without consulting a licensed physician or pharmacist
- Any discrepancy between the information provided and the product's package insert or SFDA guidelines
- Any misuse of medication resulting from personal interpretation of the content displayed
Important notice: Drug formulations and instructions may vary between production batches. Always rely on the leaflet included inside the product packaging you have, and consult your pharmacist or physician before starting, adjusting, or discontinuing any medication.
By using this content, you acknowledge that you have read this disclaimer and agree that Al Mujtama Pharmacy bears no liability arising from reliance on this information as a substitute for direct medical consultation.
Your health is a trust - always consult your doctor first.
-1744229570.gif)